Authors / metadata
DOI: 10.36205/ trocar3.2022001
Abstract
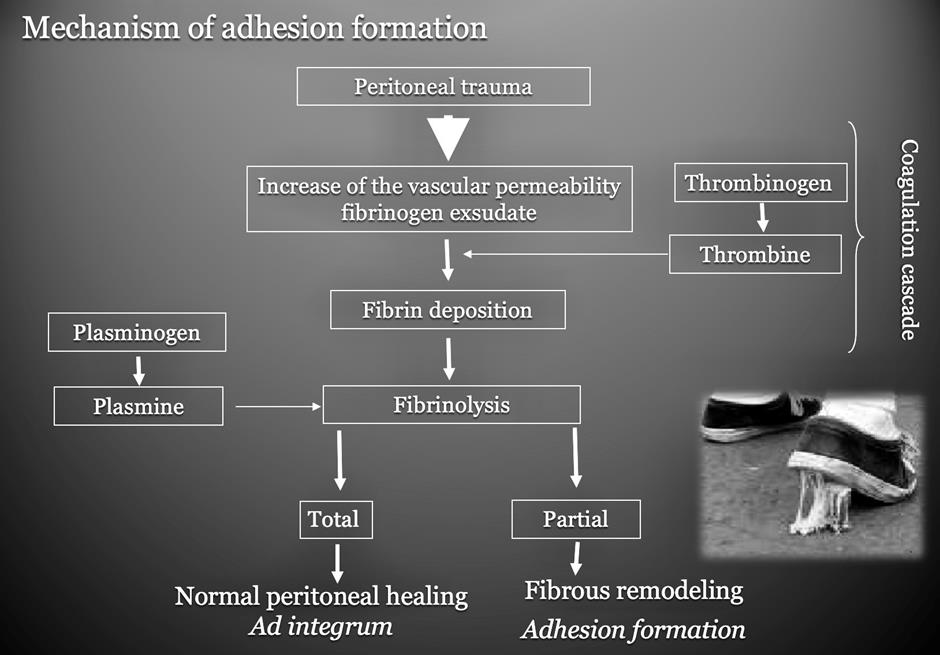
Manipulating tissues during surgical interventions causes adhesions. Some tissues and some patients are more prone to form adhesions. When cross linked fibrin is not broken down by the activation of plasminogen to plasmin there remains a fibrin clot causing the adhesion (Tab 1). The incidence of adhesions in open surgery is estimated at an average of 85% in literature. Readmission due to Adhesion Related Disease (ARD) (1) is evaluated at 5% – 10%. Laparoscopy is comparable to laparotomy. Gynaecological surgery patients are seen back by the gynaecologist only in 52% of cases, 26% is seen in general surgery and 14% in urology. Surgery of the ovaries is most at risk (48,1%) followed by fallopian tube surgery (41,2%), uterine surgery (32,1%). Endometriosis surgery has an adhesion incidence of 82% followed by tubal surgery and adhesiolysis 76%, ovarian surgery 75% and myomectomy 68%. Recent investigations have suggested that good surgical technique combined with adhesion barriers does reduce the incidence of adhesions. In laparoscopy and laparotomy meticulous surgical technique is very important. All residual blood has to be rinsed off, preferably with Ringer’s lactate combined with the appropriated amount of heparin, diluted and aspirated this after meticulous haemostasis. When possible monopolar and bipolar energies should be replaced by ultrasound or laser (CO2 laser) energy. Using floatation barriers seems not to have an influence on adhesion formation. Gel barriers have a significant effect on adhesion formation as documented by second look laparoscopies. Sheets do prevent adhesions. Using NSAID and corticosteroids in preventing postoperative pain as to help early ambulation after surgery are mainstay in surgery and should be endorsed in the policies to prevent adhesions.
Introduction
Adhesions and its synonym synechiae (Greek for continuation) are constituted of fibrous band between organs and are one of the underestimated problems complicating the postoperative phase of surgery (Fig 1). Liakakos et al found adhesions to be present in more than 90% of patients after open abdominal surgery and in 55% – 100% of women after pelvic surgery.
The trauma inflicted to the peritoneum starts the process leading to adhesion formation (2) (Fig 2 a-b). In a large study Ellis et al on 29.790 patients,found that 35% of the patients had to bereadmitted, twice on average, to the hospitalafter open abdominal or pelvic surgery due toAdhesion Related Disease (ARD) (1). More than22% of readmissions did occur within the firstyear after the initial surgery. The Surgical andClinical Adhesion Research study (SCAR) stated that the readmission rate over a period of ten years was 5% for open surgical procedures in gynaecology (3). 40% of the patients with ADR have been readmitted two to even five times. The overall number does suggest that a great number of adhesions do not cause clinical symptoms.
Intra Uterine Adhesions (IUA) are strings of fibrous tissue connecting opposite walls of the uterine cavity. IUA are found after control hysteroscopy in 31,3% after myomectomy, 6,7% after septectomy and 3,6% after polypectomy (4) There is a conundrum in denominations Asherman’s syndrome (Joseph Asherman 1889 – 1968) (5). He does refer first to a complete occlusion and stenosis of the cervical os, with as consequence amenorrhea but with atrophic endometrium in the cavity whilst the second syndrome is IUA characterized by painful, reduced menses and secondary infertility. Both syndromes are described after trauma in the uterine cavity under influence of pregnancy hormones. In both syndromes there is a dysfunctional endometrium more pronounced in the first syndrome (Mark Hans Emmanuel, personal communication 2021) The time to
complete intrauterine wound healing is patient dependent and depends on the type of surgery. Full postoperative, healing of the uterine cavity was achieved after hysteroscopic polypectomy in 86% (23/37 women), adhesiolysis in 67% (30/45 women), metroplasty 19% (3/16 women) and myomectomy 18% (12/65 women p<0.05). Metroplasty seems to cause more de novo adhesions 88% (3/16 women) and adhesiolysis 76% (33/45 women) as compared to myomectomy at 40% (26/65 women) or polypectomy at 0% (0/37 women). Overall women with de novo IUA did have difficulties in achieving complete endometrial integrity compared to women with no de novo adhesions 31% (23/74 women) versus 61% (54/989 women) P=0.0003. Yang et al conclude that complete healing is achieved in a range from one to three months following polypectomy and myomectomy (6).
ISGE suggests to use these data to discuss the IUA problem with the patients undergoing hysteroscopic surgery. (Recommendation II-1/A)
Clinical significance of post-operativeadhesions
A)Laparoscopy
The complications will depend on the organs afflicted by the adhesions meaning the location of the adhesions in most cases these will cause chronic pelvic pain, bowel obstruction and infertility (2 – 3)
The complexity of abdominal entry in an ARD abdomen adds significant risks to subsequent surgical reinterventions (7-8)
B)Hysteroscopy
IUA are symptomatic through menstrual abnormalities (amenorrhoea, hypo-menorrhea, Irregular bleeding). In patients taking oral contraception and/or hormonal therapy these symptoms can be masked. The latter are also masking possible pelvic pain and dysmenorrhea. Reproductive outcome is poor the prevalence is estimated at 43% (922/2151 women) (9). Recurrent miscarriages occur in 5% – 39% of women with IUA (10). An unexpected but very serious problem are the obstetrical complications like placenta accreta/increta, risk of preterm delivery, peri partum hysterectomy due to uterine rupture (11).
Pathophysiology
A)Hysteroscopy
Interference with the endometrial lining both by blind procedures like tissue sampling and or curettage, especially post abortion or postpartum (90%) (12) and hysteroscopic procedures can cause IUA. Genital tuberculosis does create IUA but the etiologic importance in the creation of IUA by other forms of endometrial infections is to be proven. The mechanism of restoration of the endometrium are still poorly understood (13).
B)Laparoscopy – Laparotomy – Gaslesslaparoscopy
I.Classical Model: local phenomenon betweenopposing lesions
Damage to peritoneal surfaces induces a response starting with an acute inflammatory reaction, and a process involving mesothelial cells, macrophages, exudate with cytokines and coagulation factors, neutrophils and leukocytes (14).
Within hours a peritoneal defect (i.e., caused by a trauma during surgery) is covered with macrophages and mesothelial cells (15). In the circumstances that mesothelial cells are able to cover the lesions fibrinolysis will start and after a few days this will result in re-epithelialization and a smooth surface without IUA. When fibroblast do invade the fibrin clot these will start to grow and proliferate and start to form IUA.
II.Adapted model: the role of the peritonealcavity (16).
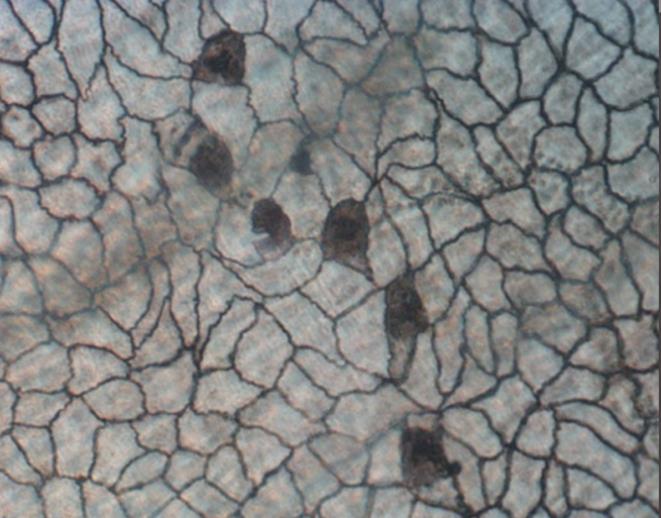
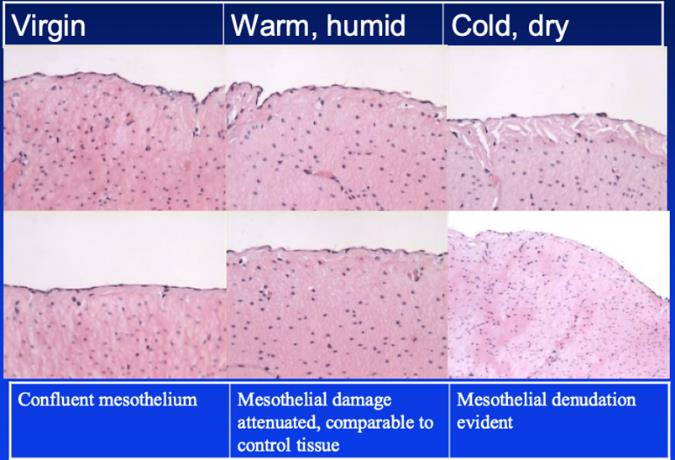
Free floating mesothelial cells are present at all times and their number increases twelve times within in two to five days after injury (17). When extensive lavage is performed at the time of surgery a substantial number of these cells are removed slowing down the natural healing (18). CO2 gas is used to distend the abdominal cavity in laparoscopy this does interrupt homeostasis the same with room air in laparotomy and gasless laparoscopy influencing the mesothelial cells (Tab 2). The direct relation between CO2 insufflation, acidification of the peritoneum and decreased immunoprotection might thus result in an altered adhesion formation (19) (Fig. 3–4). Identified so far is hypoxia of the mesothelial cells due to CO2 pneumoperitoneum. Desiccation of cells, by CO2 pneumoperitoneum and tissue manipulation, have been demonstrated to increase adhesions and this increase is time- and pressure-dependent (20-21).
In gasless laparoscopy care has to be taken not to compromise the anterior peritoneum by pressure of the lift system (22).
Prevention of adhesions in hysteroscopy
A)IUCD
In the literature there is no evidence that IUCD prevents IUA formation. The physical barrier of the IUCD is not proven as the uterine cavity is a virtual cavity and hence separation of layers is only present at the specific position of the IUCD. This means that small T shaped IUCD’s have little to no impact. The IUCD should be very large in surface (23). The other effect of the IUCD is that, as a foreign body, it genders an inflammatory response in the endometrium responsible for a negative effect on the recolonization by the endometrium at the impaired surface (24). The progesterone IUCD’s suppress the endometrium and are therefore not to be used. The blind introduction of an IUCD after a major hysteroscopic procedure, adhesiolysis, septectomy or myomectomy represents a major risk of infection and perforation (25-26).
The only small RCT does not show difference between the use of only an IUCD and hormonal therapy or a hormonal therapy alone (27).
ISGE recommendation: there is no evidence that IUCD’s do reduce IUA formation. Stage 1: A
B)Barriers
The effectiveness of barriers, hyaluronic acid gel and polyethylene oxide–sodium carboxymethylcellulose gel, has been assessed by five randomized studies and these have been reviewed in several meta-analyses (28,29,30,31,32,33). There is no evidence for an effect favoring the use of any barrier gel following operative hysteroscopy for the key outcomes live birth or clinical pregnancy (RR 3.0, 95% CI 0.35 to 26, P=0.32, 1 study, 30 women, very low-quality evidence). The use of any gel following operative hysteroscopy decreases the incidence of de novo adhesions at second-look hysteroscopy at 1 to 3 months (RR 0.65, 95% CI 0.45 to 0.93, P=0.02, 5 studies, 372 women, very low-quality evidence). After using any gel following operative hysteroscopy there are more AFS 1988 stage I (mild) adhesions (RR 2.81, 95% CI 1.13 to 7.01, P=0.03, 4 studies, 79 women) and less stage II (moderate) adhesions (RR 0.26, 0.09 to 0.80, P=0.02, 3 studies, 58 women) or stage III (severe) adhesions (RR 0.46, 95% CI 0.03 to 7.21, P=0.58, 3 studies, 58 women) (all very low-quality evidence).
ISGE recommendation: Gynaecologist can use gel barriers as these tend to reduce de novo adhesion formation as the one formed are less moderate to severe and more mild adhesions (very low-quality evidence). Gynaecologist should council their patients that there is no evidence for better pregnancy rates or higher birth rates following the use of barriers in operative hysteroscopy Evidence and strength of recommendation I: C.
C)Medical
Randomized studies to not point at any evidence that estrogens do prevent IUA.
Since its first use in 1964 several regimens have been proposed to promote the re-epithelialization of the endometrium after adhesiolysis (34).
No data exists on the ideal dose and length of the therapy. Preoperative estrogen therapy has been suggested to optimize the endometrial growth before the surgical intervention; however, evidence on its effectiveness is lacking. Moreover, the possible adverse effects of hormonal therapy (nausea, thromboembolic disease) should be taken into account when considering its use (35).
ISGE recommendation: Gynaecologists should council their patients that the use of estrogen therapy after hysteroscopic surgery lacks scientific evidence and could gender side effects Grade II-1: C.
There is no significant evidence from any published study to recommend the use of steroids (such as dexamethasone, hydrocortisone, and prednisolone) in humans, and several side effects still have to be ascertained (36,37).
ISGE recommendation: Gynaecologists are advised to council their patients that the use of steroids cannot be recommended to avoid adhesion formation and could have side effects. Grade II-2: C.
Some case reports describe the use of other medication (aspirin, sildenafil citrate, and nitroglycerin) to promote the perfusion of the endometrium. At present no evidence exists on its efficacy and therefore its use cannot be sustained.
Surgical technique and equipment
Cold scissors: Here there are several advantages as it remains the most accessible means to deal with adhesions, further the surgeon has a direct view on the surface to treat she/he can avoid destruction of the surrounding endometrium. The five French channel allows for sturdy instruments and the small barrel hysteroscope (3.8 mm median) allows for assessing the uterine cavity without cervical dilatation. The latter allows for treatment of IUA without anaesthesia or sedation in an office setting (See & Treat).
Uni-polar electrical energy implements that the electrons released by the active electrode on the tissues have to travel through the tissues to the neutral, passive electrode hence causing collateral damage up to 0.6 mm in depth as a median and therefore does lengthen the time period to a restitutio ad integrum of the endometrial lining. Anionic distention medium is a must with possible distention fluid complications. The instruments are of 27 to 25 French outer diameters necessitating a cervical dilatation from 10 – 8 Hegar respectively hence requesting general anaesthesia. Today bivalent uni-bipolar resectoscopes of 15 French are available, not in need of cervical dilatation, but the uni-polar current is felt as painful and irritating by the patients. Here general anaesthesia is still recommended.
Bi-polar energy: the current remains the same monopolar electrical current but now traveling from the active towards the passive or recuperating pole integrated into the same instrument at some 6- 8 mm of distance in the 27 French resectoscopes. In the 15 French resectoscopes the distance between the poles is reduced as is the wattage of the current. The electrical current does travel only to the tissues to be operated under direct view. This results in less in-depth destruction of the tissues treated and the surrounding ones. For the large resectoscopes there is still need for dilatation. When bipolar needles of 5 French are used the surgeon can use small barrel diagnostic scope with an outer diameter of 3.8 mm on average. Here cervical dilatation is not necessary and as the passage of the electrical current affects only the tissues treated. The patient is less uncomfortable so office procedures without anaesthesia can be performed.
Laser Light: The only available lasers are the Nd-YAG and Diode laser systems where heat is diffused into the tissues causing thermal damage up to one cm in depth. The fibers are very fragile and expensive to replace. It can be used through small barrel scopes and can therefore be used without anesthesia or sedation. (38).
Mechanical Energy by morcellation: These instruments do not need electrical energy hence the influence on the surrounding tissues is nonexistent. This allows for quicker regeneration of the endometrial lining in polypectomy and removal of retained products of conception. (39,40).
The first generation of morcellators with an outer diameter of 26 French did necessitate general anaesthesia whereas the newer generation with an outer diameter of 19 French can be used without anaesthesia in selected, multiparous, patients for in office procedures.
Prevention of adhesions in laparoscopylaparotomy and gasless laparoscopy
A)Technical Surgical Aspects
1.Surgical manipulation
Preventing adhesions implicates the use of a meticulous surgical technique.
The aim is to adjust the technique to minimize the trauma to the different structures and above all to the peritoneum, to minimize the risk of infection, to achieve the best optimal haemostasis and to avoid contamination with foreign materials such as glove powder (41,42)
The Halstedian principles of gentle tissue handling and meticulous haemostasis prevent the presence of free blood and ischemic tissues (43). Laparoscopic approach is generally preferred over laparotomy until now however there is no evidence in randomized control trials concerning the formation of adhesions.
2.Blood
There is a lack on randomized control trial concerning the importance of blood in the abdominal cavity and adhesion formation. Animal studies however indicate that blood left in the abdominal cavity at surgery is a factor inducing adhesion formation (44). The abdomen should be cleaned with Ringers lactate at the end of the surgery. The blood is easier aspirated if heparin (5.000IU/lit) is added to the Ringers lactate as clot formation is reduced. When there are no peritoneal injuries in animals, small clots do not enhance adhesion formation whilst large clots do (45).)
ISGE recommendation: There is good evidence, in well-designed case-control studies in animals, that rising blood out of the abdomen decreases the formation of adhesions. Grade II-2: A
3.Meshes and Suture material
There are no randomized trials comparing neither meshes nor threads to the formation of adhesions in the human. Leaving a mesh exposed to the abdominal cavity (not covered by peritoneum) will result in an increased risk of adherence to the mesh, with the risk of bowel obstruction. This is also true for exposed barbed sutures (46). The presence of suture material and tightening the sutures to the point of ischemia promotes adhesion formation (47).
ISGE recommendation: gynaecologists should cover all meshes and barbed sutures. The use monofilament resorbable sutures is recommended. Grade II-1: B.
4.Equipment
Ultrasonic and laser energy can be used to minimize tissue damage. Uni-polar energy does most likely cause more tissue damage due to the passage through the tissues on the way to the return electrode.
B) Conditioning the peritoneal environment
The CO2 gas used to insufflate and to maintain the pneumoperitoneum affects the whole abdominal cavity. There is evidence from small trials in human that switching to a mixture of carbon dioxide with 10% nitrous oxide and 4% oxygen can decrease adhesion (48). A trial study on 44 women who did undergo surgery for laparoscopic resection of endometriosis demonstrated a significant reduction in adhesion formation (P<0.0005) however the patients in the study group did also receive dexamethasone, a rising with heparin was performed and the humidity and the temperature of the abdomen were also monitored (49).
C)Local products
1.Floatation barriers (Ringer’s lactate, Saline,Hartman’s solution)
Meta–analysis of clinical trial demonstrates clearly that the effect of the ‘flotation agents” is nonexistent in preventing adhesion formation both in laparotomy and in laparoscopy (50,51).
This is due to the rapid absorption of the liquids by the peritoneum at a rate of 30-60 ml/h leading to the disappearance of the fluids within 24-48 h. This time span is too short to be effective in preventing adhesion formation.
ISGE recommendation flotation barriers should not be used to prevent adhesion formation. Grade II-1: D
Adept (4% icodextrin solution – Baxter Biosurgery, Baxter International, Deerfield Il, USA) used in animal and peritoneal dialysis seems to have sufficient long intra peritoneal presence to prevent adhesion formation (52). Standard recommendation is to use the solution throughout the surgery and leave 1.000 ml in the abdomen at termination of the surgery (53,54). One randomized controlled pilot study where lavage during surgery and instillation of 4% icodextrin was practiced showed the treatment to be well tolerated and above all did show a reduced incidence in adhesion formation both in extent and severity at laparoscopic adnexal surgery however the group sizes were not powered to statistical significance (54). More recently these findings have been confirmed in a study concerning patients operated for adhesions (55). As adverse reactions are listed: vulvar edema, allergic reactions (allergy to starch based polymers and iso-maltose intolerance), fluid leakage through the wounds and in some cases abdominal distention and discomfort (56).
ISGE recommendation: 4% icodextrin solution can be used, in the prescribed methodology, in patients operated for ADR. Grade I: C.
2.Gel barriers
Hyaluronic Acid
In a large multicenter randomized trial Intergel® (ferric hyaluronate, Ethicon – J&J, Somerville, New Jersey, USA) was proved to be effective in reducing the extension and severity of post-operative adhesions as compared to Ringer’s solution in patients undergoing peritoneal cavity surgery by laparotomy with a planned second-look laparoscopy (57).
Due to unacceptable post-operative complications the gel is no longer available (58).
Hyalobarrier Gel Endo (Auto-cross linked hyaluronic acid, Nordic Pharma, Dublin Ireland) is suitable for use to prevent adhesion formation in abdominal surgery due to its adhesivity and prolonged stay in time on the treated surfaces. Thirty-six patients, treated with Hyaolobarrier Gel Endo in a randomized controlled study, who underwent laparoscopic myomectomy showed a significant reduction in postoperative adhesion formation (59). The same authors did demonstrate the application of the gel in cases of laparoscopic myomectomy to be associated with an increased pregnancy rate (60). These findings including a favorable safety profile have been confirmed in a blinded, randomized, multicenter study provided the gel is used at the end of the surgery as rinsing is able to remove the gel (61).
ISGE recommendation there is sufficient evidence to recommend the use of Hyalobarrier gel after laparoscopic surgery for myomata. Grade I: A
Solution of hyaluronic acid, Sepracoat® coating solution, a solution of hyaluronic acid, (HAL-C; Genzyme Corporation, Cambridge, USA), is a liquid composed of 0.4 % sodium hyaluronate in phosphate buffered saline, it is applied intraoperative, prior to the dissection, to protect peritoneal surfaces from indirect surgical trauma or post-operatively to separate surfaces after they are traumatized. There are no studies in the literature evaluating Sepracoat® in preventing adhesions following laparoscopic gynecological procedures. The efficacy in laparotomy was well established (62).
ISGE recommendation there is enough evidence to recommend Sepracoat in open surgery. Grade I: A
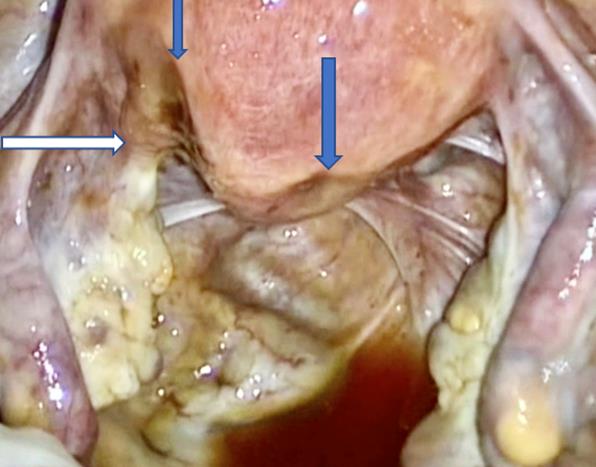
Hydrogel, Spraygel® or Sprayshield® (Covidien, Dublin, Ireland) consist of two synthetic liquid precursors that, when mixed, rapidly cross-link to form a solid, flexible, absorbable hydrogel. The solid polymer is easily applied by laparoscopy; however, it can only be applied when in contact with room air after evacuation of the CO2 this could cause air-embolisms. Sprayshield® sprayed over the affected area and remains for approximately 5 to 7 days. There after it is degraded and absorbed. One of the components contains a blue food colorant, so there is an intraoperative visualization where the gel is used. The currently available evidence does not support the use of Sprayshield® either decreasing the extent of adhesion or in reducing the proportion of women with adhesions. In a prospective blinded observational study our group, Bruno Johan van Herendael, Benedikt Tas, Marc Francx and Bart De Vree at Ziekenhuis Netwerk Antwerp, 2004 -2005 evaluated 22 patients treated with Sprayshield with second look laparoscopy compared to 22 non treated, comparable patients. The conclusion was that using the gel did reduce de novo adhesions at second look laparoscopy as compared to the non-treated patients but the study is too small to draw conclusions (Randomised observational prospective trial: never published) (Fig 5). Mettler et al randomized 64 women undergoing a myomectomy by laparoscopy or laparotomy. Only 22 returned for a second look laparoscopy. Although the treated patients were more adhesion free at second look laparoscopy compared with the control group; the difference was not significant (63). More randomized and especially blinded prospective studies are need to obtain scientific proof of the efficacy of Sprayshield®
Other gel barriers
Intercoat® (Ethicon – J&J, Somerville, New Jersey, USA) a viscoelastic absorbable gel of polyethylene oxide and carbomethylcellulose is stabilized by calcium chloride. At the end of the procedure the coating is applied in a single layer to function as a mechanical barrier. Lundorff et al published the results of a randomized third party blinded multicenter European trial showing that visco-elastic gel did significantly reduce adnexal adhesions in patients undergoing gynecological laparoscopic surgery (64).
At the same time period Young et al published a randomized study evaluating Oxiplex/AP gel (FzioMed, San Luis Obospo, California) Their conclusion is that the gel is safe, easy to use during laparoscopic surgery and contributes to a reduction of adnexal adhesions (65).
CoSeal (resorbable hydrogel polyethylene glycolpolymer solution – Baxter Biosurgery, Deerfield, Il USA) is extensively used in vascular surgery reconstruction operations (over 200.00 patients since 2002). In both open and laparoscopic surgery, provided good surgical technique is used, the gel reduces significantly, incidence, extent and severity of postoperative adhesions (66).
ISGE recommendation: There is evidence that both other gel barriers are safe to use and do reduce adhesion formation. Grade II-2: A
3.Sheets
Gore-Tex Surgical membrane (W.L. Gore & Associates Inc, Flagstaff, USA) The expanded polytetrafluorethylene non absorbable barrier is characterized by a microscopic structure preventing cellular ingrowth at the same time it is non-inflammatory. In patients undergoing laparotomy for adhesiolysis or myomectomy the membrane decreases the severity, extent and incidence of adhesions (67). The limitations of the membrane are that it has to be sutured in place and removed during subsequent surgery. It is therefore very difficult to use it at laparoscopic surgery.
Interceed (Ethicon–J&J, Sommerville, New Jersey, USA) The oxidized regenerated cellulose membrane is widely used to prevent adhesions at surgery as it has been proved to reduce adhesion formation in animals and in human. The damaged peritoneal surfaces are covered with the membrane that transforms itself to a gelatinous mass. In doing so it forms a physical barrier that separates adjacent damaged peritoneal surfaces. Over 13 clinical trials including 600 patients have been performed. The meta-analysis of 10 randomized control trials reported 24.2% reduced adhesion formation on the side treated with the barrier (68). However, in the presence of bleeding the sheet seems to aggravate adhesion formation rather than reduce it.
Seprafilm (Genzyme Biosurgery, Bridgewater, USA) this sodium hyaluronate – carbomethylcellulose membrane ids placed over a suture or an injured peritoneum without stich and remains there for some seven days. Here a bleeding seems not to impair the efficacy of membrane. In general surgery several studies did report the efficacy of Seprafilm over 20 studies on over 4000 patients are reported in the literature (67). The problem in laparoscopy is that the membrane is fragile and difficult in use.
4. Drugs
The NSAID ketorolac has shown some evidence to prevent adhesions in animals (69). Dexamethasone combined with noxythiolin (peritoneum antiinfection agent) have been tested in 126 patients operated by microsurgery and assessed by a second look laparoscopy three to six months later. In the control group 19% of the patients became pregnant versus 40% of patients in the treated group (p<0.02). The improvement in the adhesion score in the treated group was 23.3 and in the control group 10.2 (70).
Discussion
The general consensus is that adhesion formation is a persistent companion in surgery sometimes leading to ARD. To reduce ARD to a minimum good surgical technique is the key ingredient to maximal reduction of postoperative adhesion formation. It is important to adhere to Evidence Based Medicine (EBM). EBM being the integration of individual clinical expertise, best available external clinical evidence and last but not least patient’s values and expectations. Individual clinical expertise boils down to good surgical technique and the fact that it could be better to consult a colleague with expertise in ARD and send the patient to his/her center eventually to go and assist the colleague to get personal expertise. In ARD due to postoperative adhesion formation, it is important to adhere to the second pilar of EBM and integrate the best available external clinical evidence in our judgement as to help the individual patient the best possible way at the least costs for the health system. Although EBM is never sufficient to make clinical decision, EBM points to a hierarchy of evidence so that the individual surgeon can make her/his specific clinical decision. The patient’s values and expectations can be multiple but in essence these will be functional solutions to the patient’s problem be it infertility or abdominal discomfort and pain. This does not mean an anatomical solution but rather a functional solution to the problems at hand.
In hysteroscopic surgery the data suggest that mechanical tools, scissors and morcellation, interfere the least with the endometrium. In the prevention of IUA only an in office second look hysteroscopy after 6 weeks in the immediate postmenstrual period is congruent with the above EBM principles. Postoperative hyaluronic gel application can be of help but is quite expensive and at this moment in time there is no evidence. Estrogen in whatever dosage can be given especially in patients with proven hypotrophic endometrium at biopsy. There is no proof of its efficacy in hormonal balanced pre-menopausal patients.
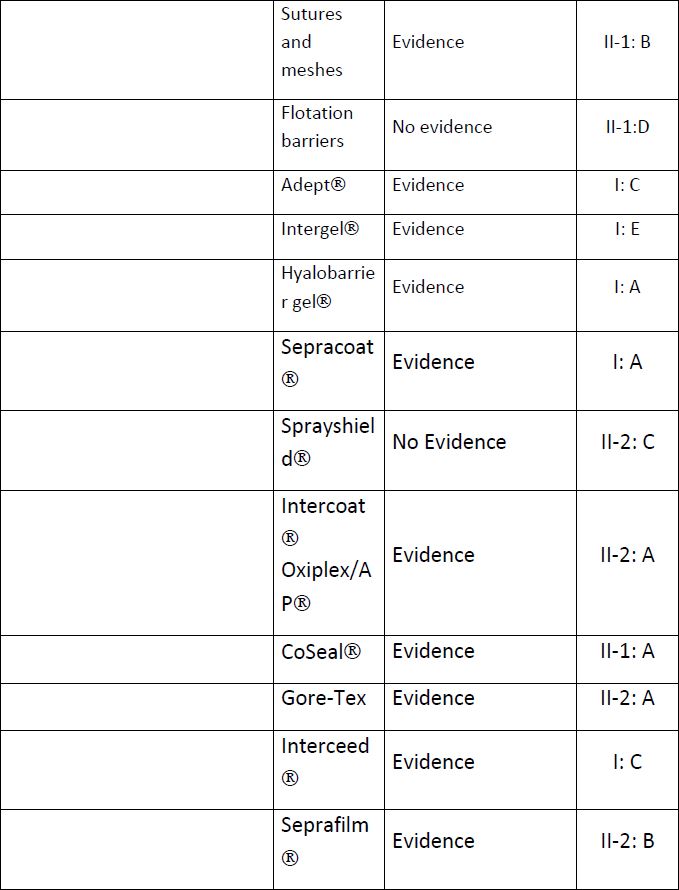
Concerning open surgery, laparoscopic surgery and gasless laparoscopic surgery meticulous surgical technique is of utmost importance. Residual blood should be avoided and this can be obtained by careful hemostasis and rinsing with Ringers Lactate with heparin. The proper sutures should be used and preferable no braided sutures are left in the abdominal cavity. The use of floatation barriers does not seem to add substantial benefit in the prevention of adhesions. Gel barriers (Hyalobarrier gel Endo® or Intercoat®) based on hyaluronic acid are proven to have a significant effect on adhesion prevention. The inconvenient is that these gel barriers are costly. We therefore do advocate a very cautious and proper use of these barriers. As for sheets there is enough evidence, they prevent adhesions. The use of NSAID in the prevention of pain and or corticosteroids in the prevention of post-operative nausea is already mainstay after surgery and can be further endorsed in the prevention of adhesions. A resume of the above reflection can be found in table 2.
Altering the laparoscopic gas to a mixture of carbon dioxide + 10% nitrous oxide + 4% oxygen may be a future option as this is an easy way to prevent adhesions, but further studies are needed to provide stronger data regarding efficacy and safety.
References
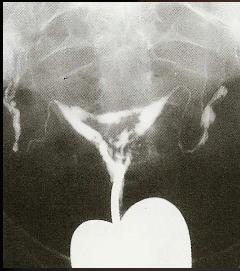
Fig 1: The classical image of Intra Uterine adhesions (synechiae) at hysterosalpingography
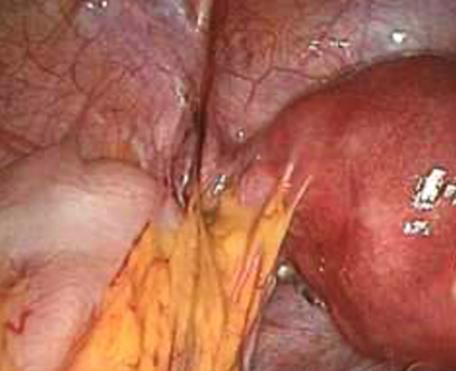
Fig. 2 Classical appearance of post operative adhesions, due to peritoneal damage by previous laparoscopic fertility surgery, at second look laparoscopy
Tab 1 The Coagulation cascade in a nutshell
Fig 3 Scanning electron microscopy of the simple squamous epithelium at 37° Celcius and relative humidity of 95%. Visible are the free floating mesothelial cells (dark entities) (Douglas E Ott)
Fig. 4 Scanning electron microscopy of the peritoneal damage caused by CO2 flow at 10 lit/min direct exposure. The 60 protective peritoneal fluid film has been blown away and the relative humidity has been reduced together with the reduction in temperature. The combination of these factors does cause the monolayer of squamous cells to dry out. (Douglas E.Ott)
Tab 2 The appearance of the peritoneum, squamous cell layer, loose connective tissue and mesothelium under different circumstances. Courtesy of Douglas E. Ott Mercer University
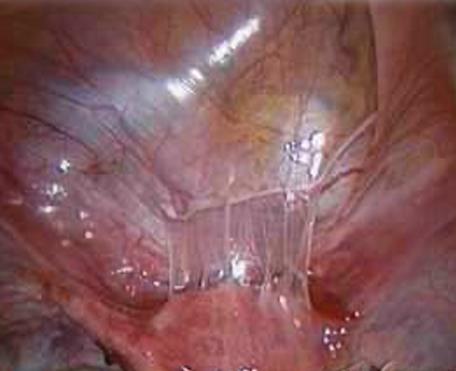
Fig 5: Second Look laparoscopy: White arrow subtle thin adhesions after major posterior myomectomy after the use of barriers (Sprayshield). Blue arrows: impressions of the ischemic sutures at laparoscopic myomectomy using Vicryl 1 resorbable suture. (ZNA series second look)
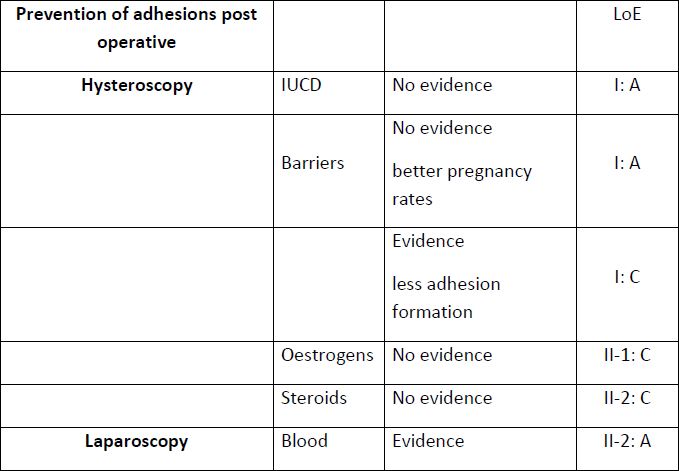
Tab 3: ISGE recommendations to prevent adhesions in hysteroscopy and laparoscopy, open surgery and gasless laparoscopy
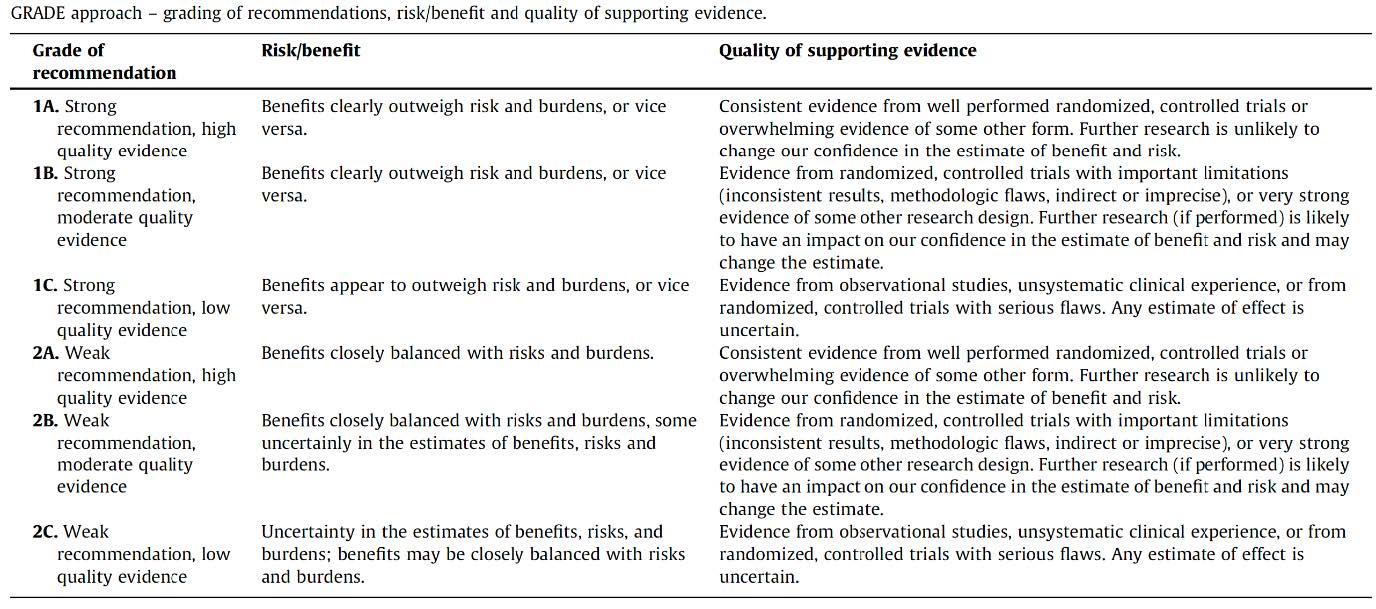
Tab 4 Recommendations Different grades and their explanation